-
QuidelOrtho: Quality
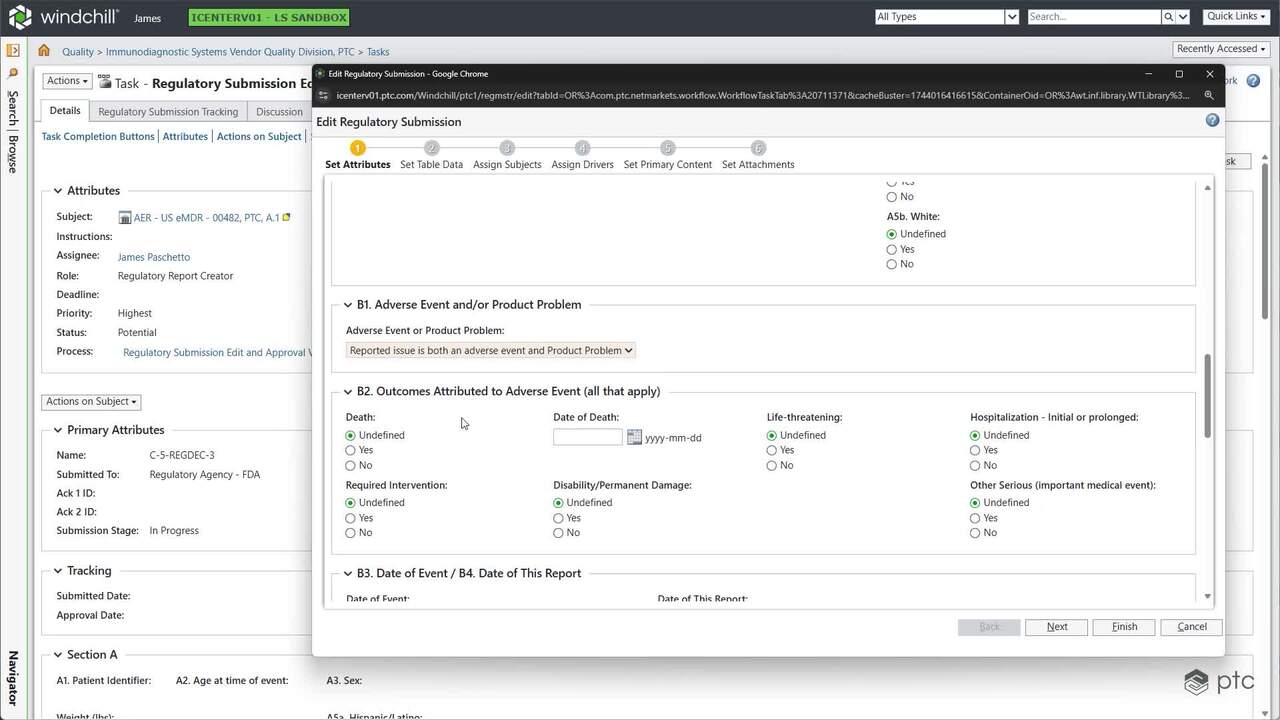
With Windchill enabling design controls and quality management to function in the same space, MedTech companies can maintain the highest quality standards, while lowering the cost of quality and ensuring compliance, traceability, and product safety.
12:55
-
QuidelOrtho: Change Management
With unified change management worldwide, MedTech companies can reduce the number of changes; optimize running costs, SOPs, training, and support staff; and accelerate time-to-market by up to 20% by cutting design redundancies.
27:53
-
QuidelOrtho: Innovation
PTC's Intelligent Product Lifecycle solutions enable a data-centric approach that supports end-to-end digital traceability for compliance. Harmonizing and focusing design cycles around innovation, manufacturing, and regulatory approval.
14:23
-
Fresenius: Medical Software Driven Innovation
See how Fresenius is driving medical technology forward by leveraging PTC's software and the concept of a closed-loop digital thread.
5:31
-
Fresenius: Medical Closed-Loop Quality Workflow
Quality is of the utmost importance when manufacturing and deploying medical devices. Fresenius uses PTC software and a closed-loop approach to ensure their products are built and operated at the highest standards.
5:51
-
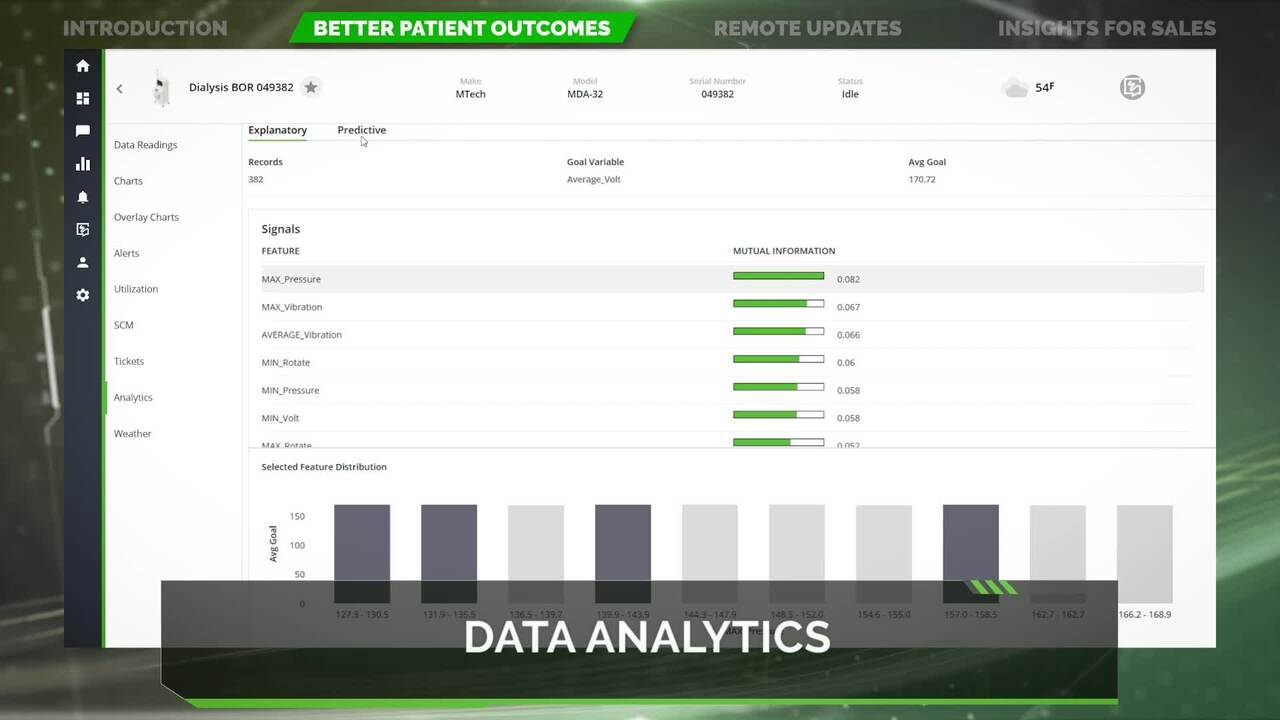
Fresenius: Medical IoT
The Internet of Things has opened up new possibilities when it comes to medical devices and their capabilities. See how Fresenius and PTC together can create better patient outcomes by leveraging IoT.
4:44
-
Quality, Risk and Regulatory Digital Thread for Medical Devices
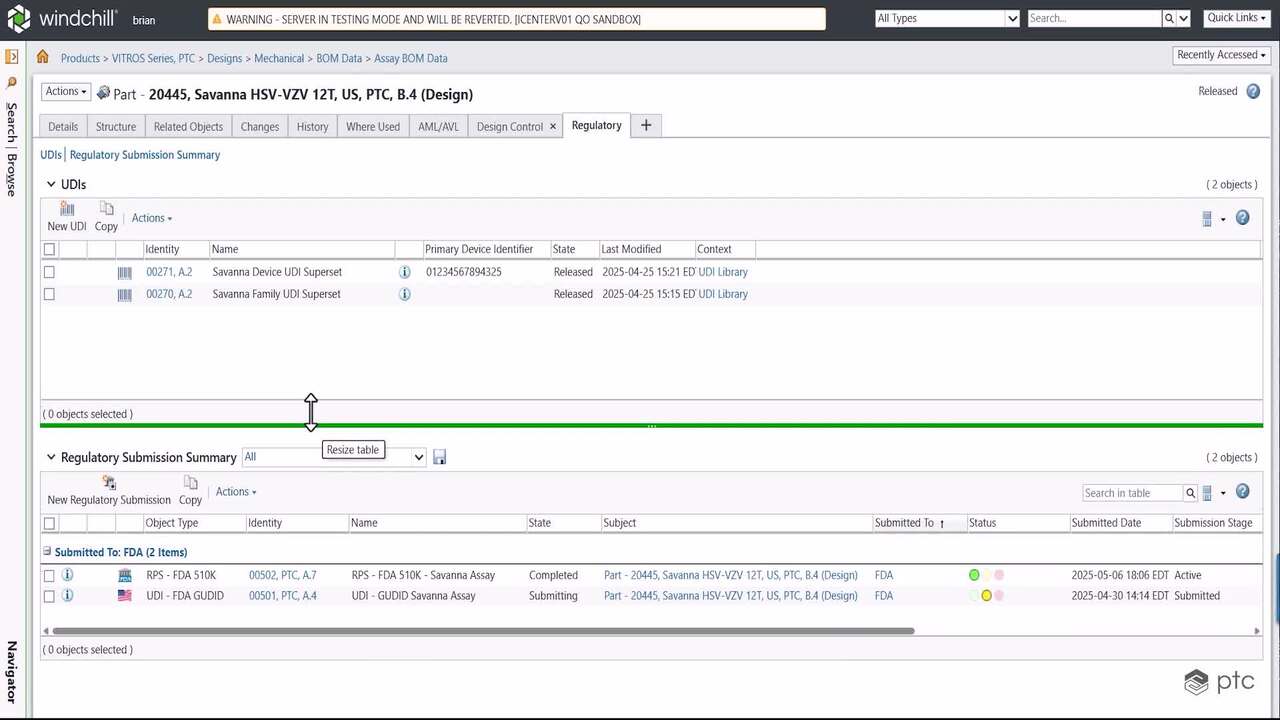
Deliver improved patient outcomes and reduce costs by building a digital thread that ensures closed-loop quality and end-to-end traceability for medical devices. Accelerate innovation and manage regulatory risk with PTC.
4:09
-
ThingWorx Connected PLM - Weaving Quality into the Digital Thread
Keeping a modern enterprise aligned is essential in today's business environment. Create higher quality products, more efficiently than ever by combining the power of ThingWorx with Windchill for better visibility, traceability, and decision making.
3:35
-
WPQ - Bill of Info
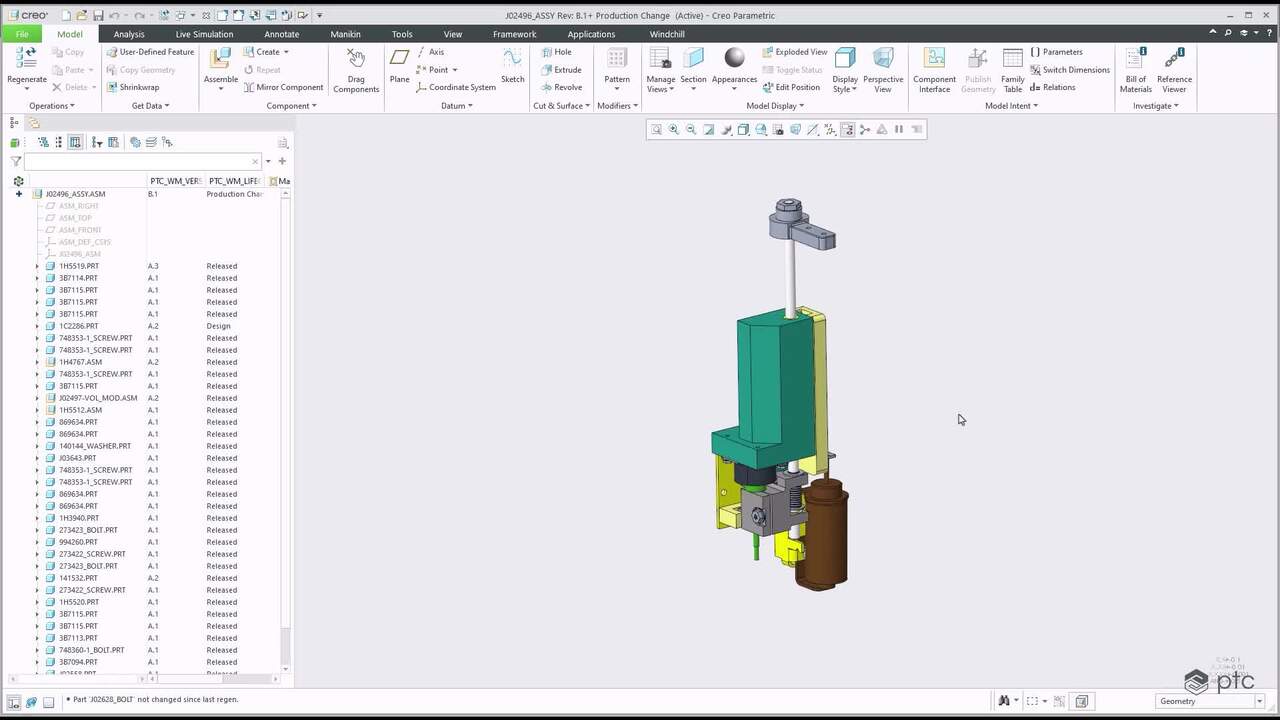
The Bill of Information provides a single source of truth on products parts, structure, and change history.
1:30
-
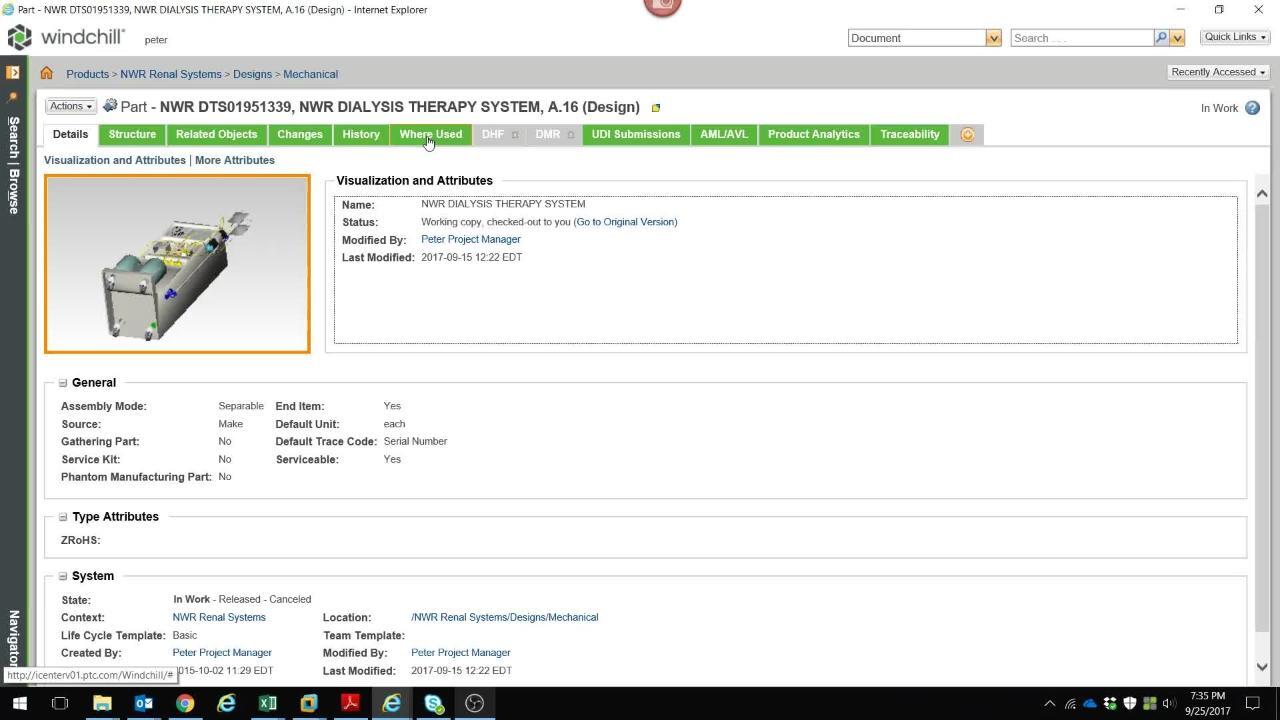
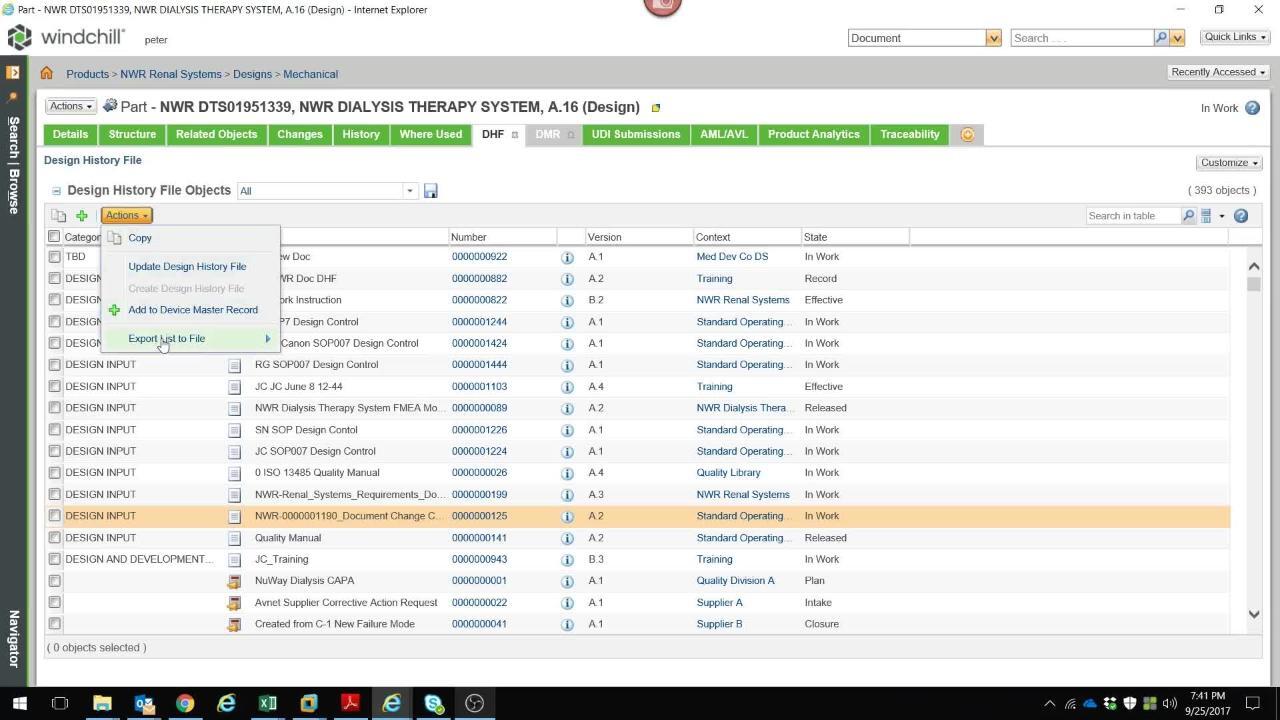
WPQ - Design Control
Effective Design Control integrates DHF (Design History File) and DMR (Device Master Record) documentation into your product realization process --resulting in significant cost and time savings, improved quality and reduced compliance overhead.
1:31
-
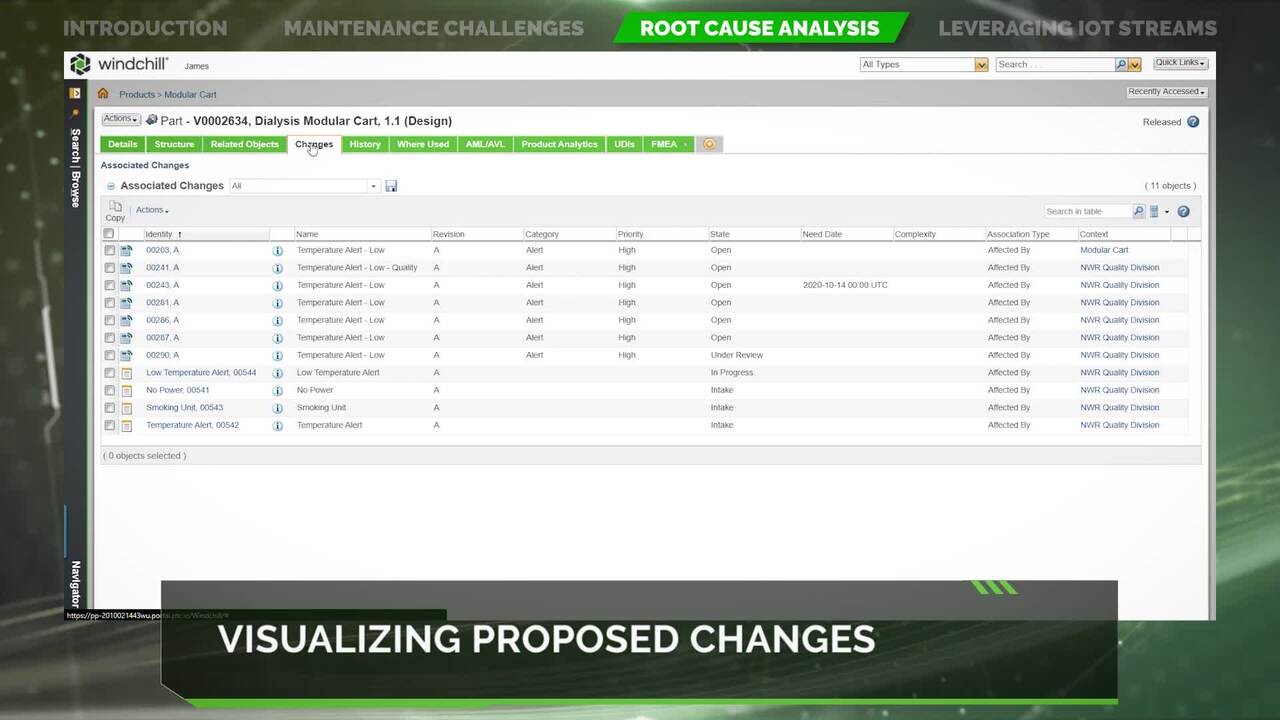
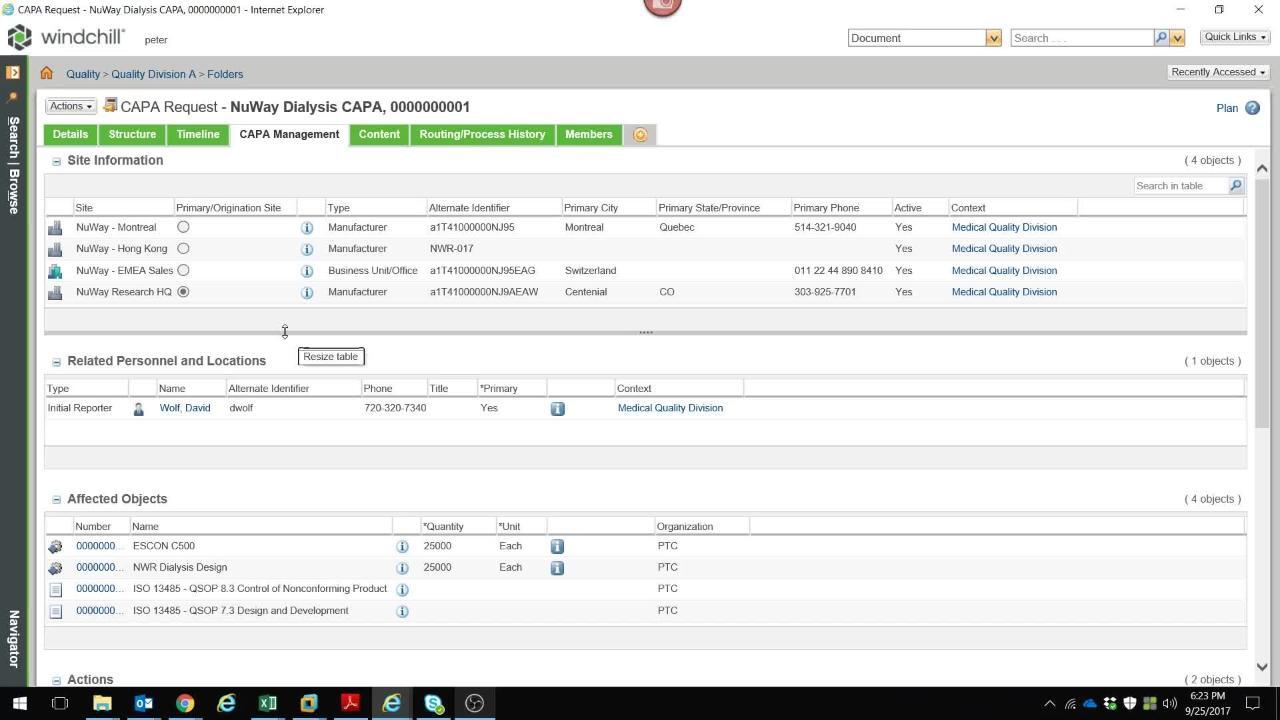
WPQ - CAPA Management
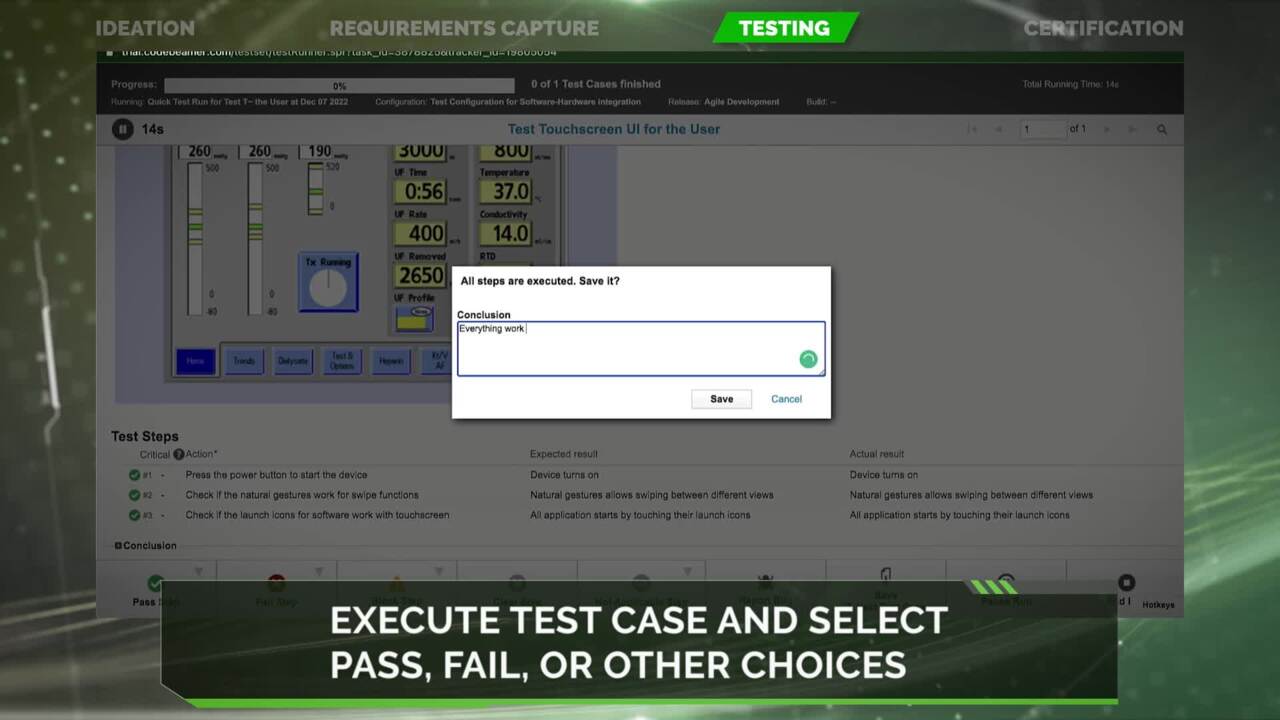
Explore a Corrective and Preventive Action (CAPA)/Supplier Corrective Action Request (SCAR) module that enables rapid investigation, root cause analysis, corrective/ preventive actions, and close-out of quality issues across the product lifecycle.
0:47
-
WPQ - Closed Loop Quality Management
Understand how the interlocking quality processes automated in Windchill Product Quality enable a robust closed-loop quality management approach.
1:56
- Load More